In 2019, I was in the middle of a research effort funded by the Israel Science Foundation to quantify the effects of external events on the structure of networks. I was invited to BGU to give a talk on my findings, and there I met Yuval Shahar. We started planning a collaboration when Covid-19 hit. During lockdowns, Yuval, an IS professor and a medical doctor, was my go-to person for understanding viruses and their transmission. Thus, in the early days of COVID-19, we started formulating the disease progression, gathering all new evidence available. We analyzed an early outbreak in a Korean church and correlated viral load with exposure duration and intensity. We analyzed death rates at first outbreaks by age group. We devised a statistical model for early estimation of the prevalence and severity of an epidemic or pandemic from simple tests for infection confirmation.
Soon after, we were designing a methodology for quantifying the interplay of viral transmission in real-world societies while considering human behavior. Covid-19 was progressing in the world, country after country. Yet, it became clear that the virus was progressing differently in different societies. The known R0 metric, describing the transmissibility of a disease, was not accurately capturing the viral transmission in different communities. Yuval and I hypothesized that the societal temporal interactions and exposure duration accounted for these differences.
At that stage, epidemiologic simulations were usually done on static models of communities. It was evident that many epidemiologic results would change under real-life conditions that take into account temporal characteristics such as temporal paths and the mathematical characteristics of real-life interactions such as the rate with which people meet, the distribution of interactions, and the duration of meetings. We were eager to model our understanding and research the interplay between real-life interactions and viral contagious using the models we devised. We wanted to explore the effect of symptomatic and asymptomatic transmission in these communities and model various distancing strategies.
During the lockdown, recruiting research assistants and students was challenging. However, upon consulting Asaf Shapira, he recommended Yanir Marmor and Alex Abbey. After a brief phone or Zoom conversation with each, I quickly realized that Asaf's recommendations were excellent, leading them to join my lab as research assistants.
In the next two years, we devised a detailed model of the disease progression in humans, which considered different progressions for asymptomatic and symptomatic individuals and considered various disease progression scenarios. Gathered Covid data from 15 countries (with the help of Yaniv Wisney); Identified and analyzed a dataset of real-world interaction data; Devised the first open-source Temporal path preserving Random Networks Generator; Devised the first interaction-driven modeling of viral transmission in societies using real-world interactions and incorporated the detailed disease progression model we developed. Our model, driven by real human interactions, linked the infection probability with the interaction duration—as is the case for airborne viruses—and incorporated human behavior dynamics with the disease progression stages in humans. This modeling enabled the prediction of outcomes at both the population and the individual level, as described below. We made the open-source code of the Interaction-Driven Contagious Model with Individual Disease Progression Modeling freely available.
In a series of works, we demonstrated that the temporal dynamics of a community have a more significant effect on the spread of the disease than the characteristics of the airborne pathogen, supporting our initial hypothesis. The research culminated in several additional interesting findings:
Effectiveness of social distancing strategies:

The various social distancing strategies employed
Evaluating different social distancing strategies, we demonstrated the superiority of decreasing daily social interactions over partial isolation strategies, such as the spatial distancing pods and a spatiotemporal distancing strategy.

We found that Social distancing strategies are effective only at the beginning of the viral spread when the vast majority of the population has not been infected yet.
These results appear in our Journal of Biomedical Informatics Publication.
The effect of the population dynamics on the spread of the virus

Highly active communities are more susceptible to viral infection. In highly active communities, slow-spreading pathogens (i.e., pathogens that require more prolonged exposure to infect) spread roughly at the same rate as fast-spreading ones. This result is surprising since slow and fast pathogens follow different paths. A strategy to overcome this is to reduce the rate of daily interactions, thus reducing the spread of the disease.
These results appear in our 2024 Journal of Biomedical Informatics Publication.
Predicting outcomes
We analyzed community and individual outcomes
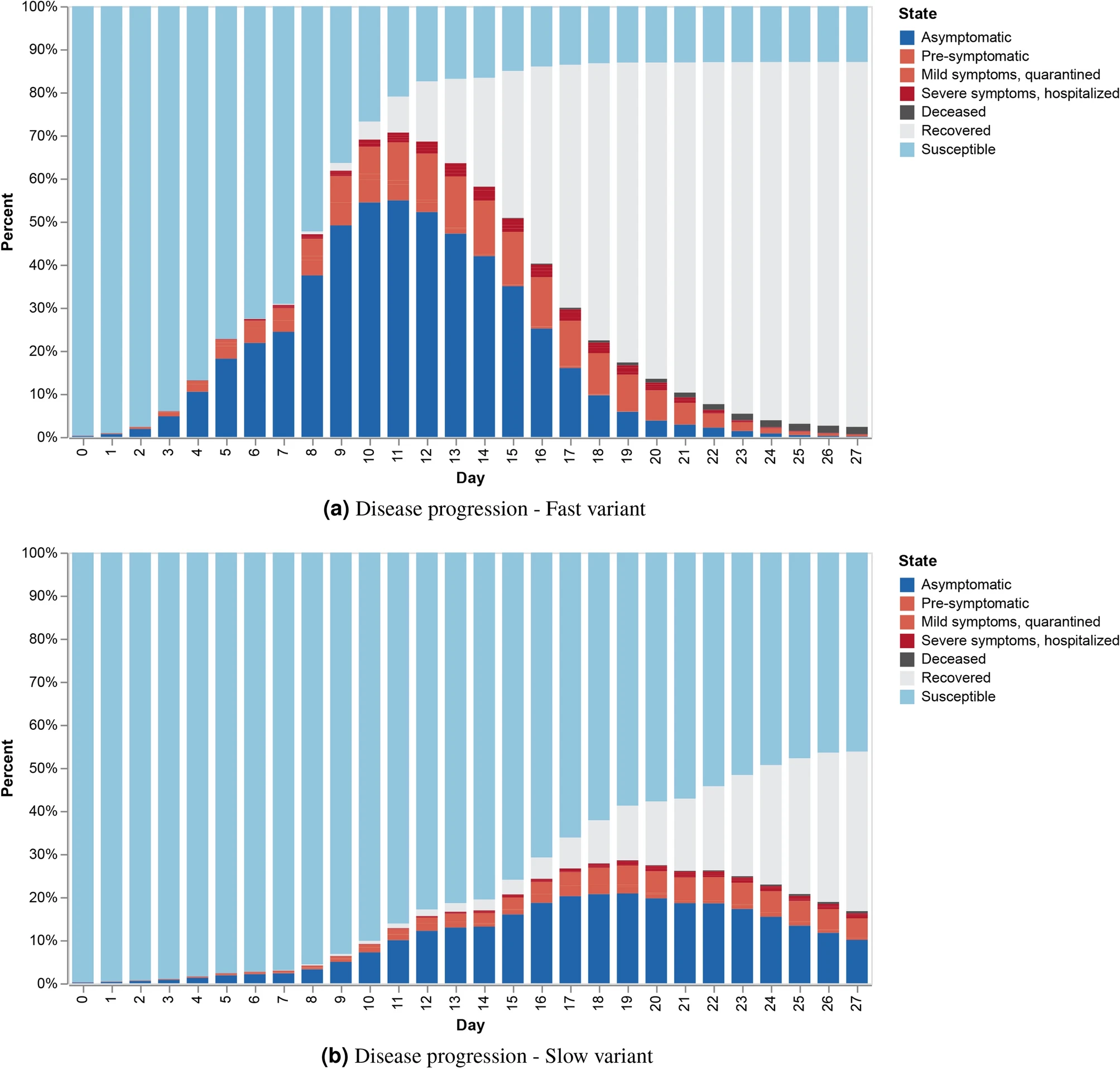
COVID-19 Disease progression with individual outcomes at the community level
We devised a prediction for the individual risk of engaging in social interactions as a function of the virus characteristics and its prevalence in the population:
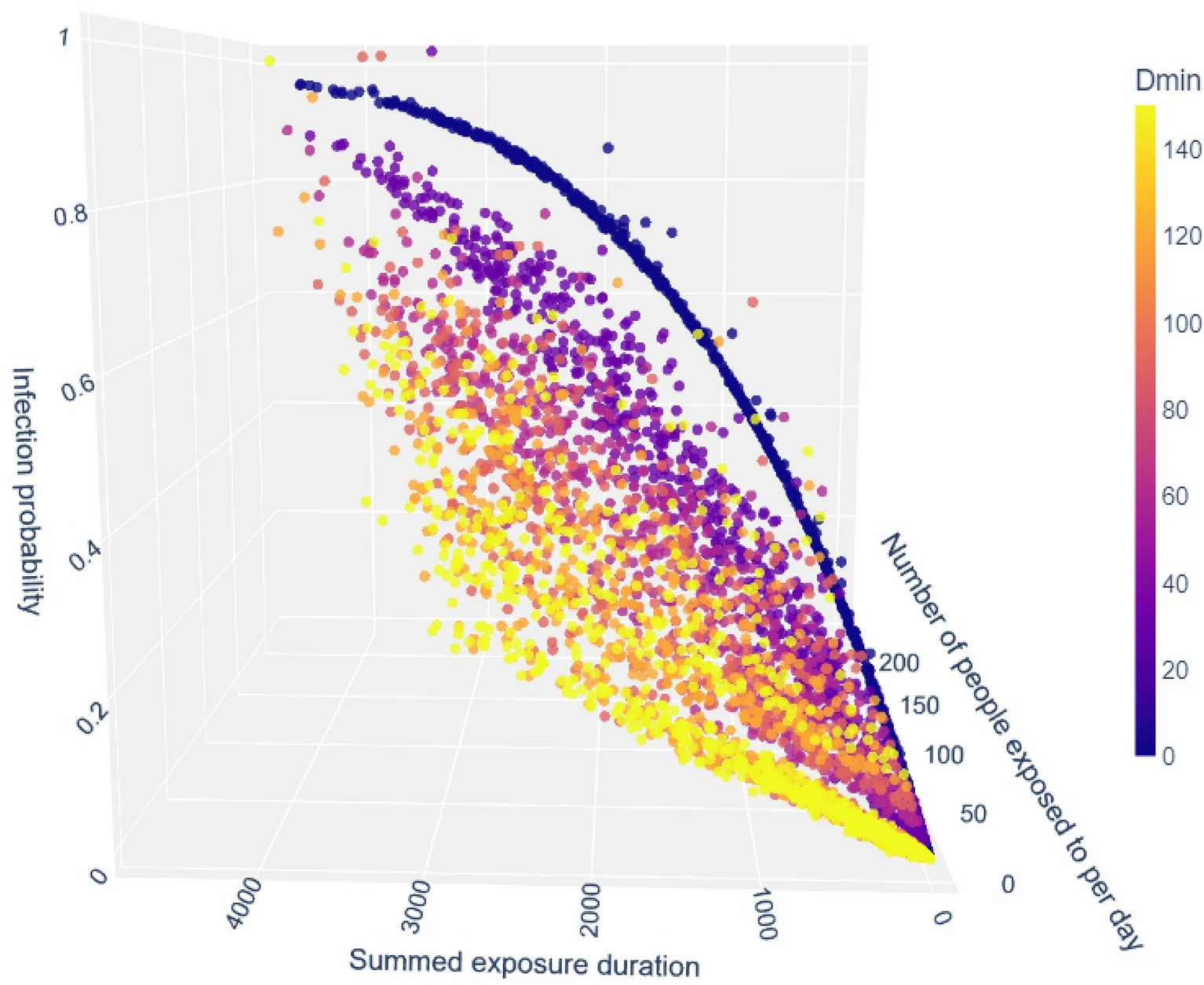
Individual probability of an adult getting infected in an average day as a function of the number of exposures and their aggregated duration, according to the model, for various exposure (Dmin) durations.
Researching asymptomatic airborne transmission, we found that asymptomatic transmission accelerates viral transmission only in sparse communities, as the enigmatic nature of asymptomatic transmission stems from the latent effect of the temporal network density on transmission.
The above results appear in our 2023 Scientific Reports Publication.
Pathogen competition conditions:
We identified the conditions under which the competition between several (two and three) competing airborne pathogens will result in the slower pathogen creating a second wave of infection that infects most of the population

We then show that when the duration of the encounters is considered, the spreading dynamics change significantly. Our results indicate that when considering airborne diseases, it might be crucial to consider the duration of temporal meetings to model the spread of pathogens in a population:
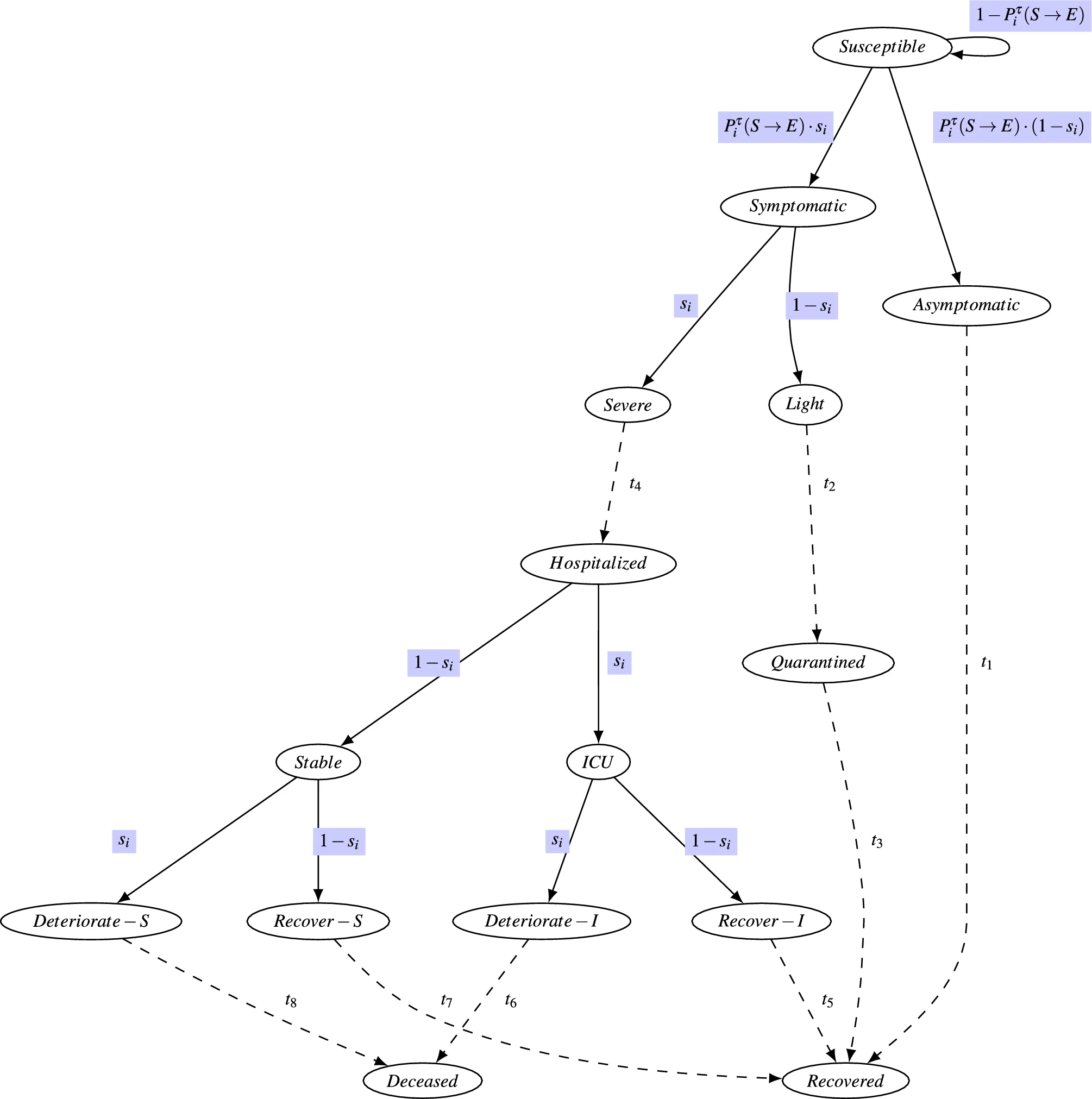
The above results appear in our 2022 Scientific Reports Publication
